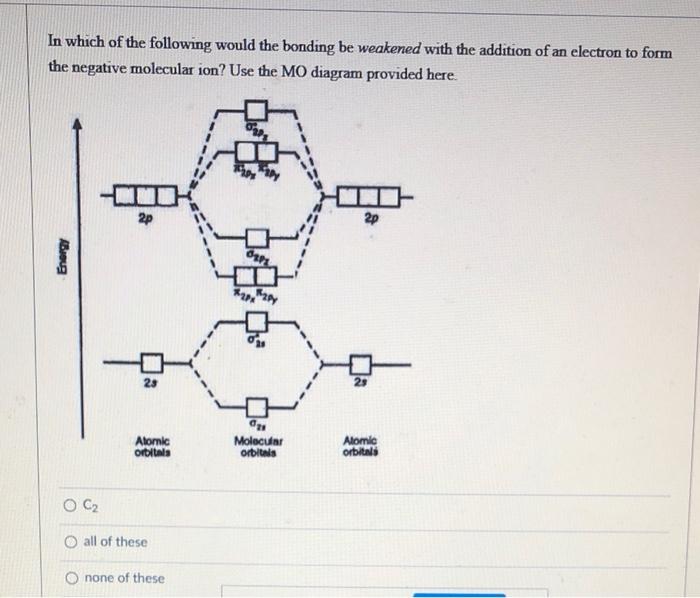
Compare the relative stability of the following species and indicate their magnetic properties: O(2),O(2)^(o+),O(2)^(ө)(superoxide),O(2)^(-2)(peroxoide).

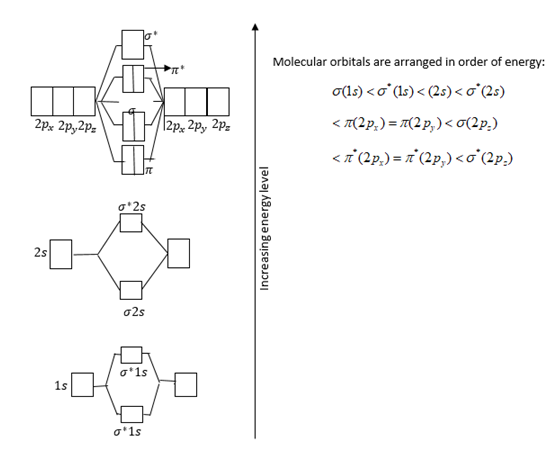
Draw and write the molecular configuration of nitrogen molecule. Claculate its bond order and magnetic property.

Effect of oxygen contamination on structural and magnetic properties of MnPd bilayer grown on Fe/MgO(001): Ab initio study | SpringerLink
Compare the relative stability of the following species and indicate their magnetic properties; - Sarthaks eConnect | Largest Online Education Community

Write the molecular orbital electronic distribution of oxygen specify its bond order and magnetic - Brainly.in
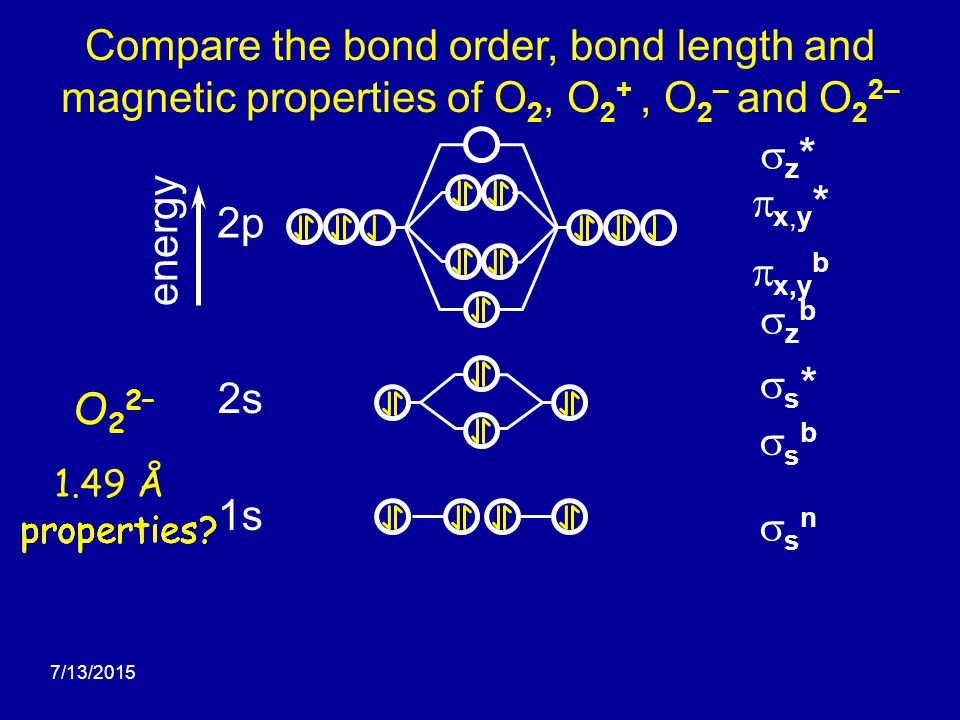
Molecular Orbital Theory Edward A. Mottel Department of Chemistry Rose-Hulman Institute of Technology. - ppt download

Effect of O2 adsorption on magnetic properties of oxygen-deficient ZnO nanoparticles - ScienceDirect
Magnetic Properties of the Oxygen Molecule in Solid Oxygen‐Argon Mixtures: Journal of Applied Physics: Vol 40, No 3

Write magnetic properties of O2+, O2-, O22-,O2 - Chemistry - Chemical Bonding and Molecular Structure - 11117029 | Meritnation.com

Write the molecular orbital electron distribution of oxygen `(O_(2))` Specify its bond order and - YouTube

Compare the relative stability of the following species and indicate their magnetic properties : O2, O^+2, O2^ - (superoxide) and O2^2 - (peroxide).

Compare the relative stability of the following species and indicate their magnetic properties: O2,O2+,O2